A wide range of natural, biodegradable and biocompatible materials have been successfully electrospun and this makes it a very attractive candidate for skin graft application. Modifications may also be made to the structure and organization of the membrane to improve its performance as skin graft. Various functionalities such as hydrophobicity, anti-bacterial property, drug release and bioactivity can be incorporated into the nanofibrous membrane to find the best combination for wound healing. Early in vivo studies using various electrospun fibers such as polyurethane [Khil et al 2003] and collagen [Rho et al 2006] on full thickness wound healing showed encouraging results although the rate of epithelialization was not reported. Kang et al (2010) did a preliminary in vivo study of wound healing on a hairless mice using heat treated electrospun polyvinyl alcohol (PVA) with and without chitosan coating (C-PVA). With a wound created using a 6 mm biopsy punch, both PVA and C-PVA showed wound contraction ratio of 90% and 91% respectively compared to 75% in the control wound (no graft) at 7 days. Histology results revealed denser collagen and thicker epidermis on the wound site treated with C-PVA compared to PVA demonstrated better healing with chitosan coating. The presence of natural polymers may introduce better biocompatibility to the scaffold.
Multiple active ingredients may be mixed together into a solution and electrospun to construct a multi-functional scaffold. Sen et al (2022) dissolved three materials, hibiscus leaves mucilage, polyvinyl alcohol and pectin (HLM-PVA-Pectin) in distilled water to form a solution. HLM has the properties of antibacterial and skin moisturizing effect, pectin has broad-spectrum antibacterial activity, absorbs and retains fluids from wound and PVA which function as a carrier polymer for electrospinning and also provides the necessary mechanical support. The resultant membrane undergoes glutaraldehyde-HCl vapor crosslinking to increase strength and maintains fiber integrity in a wet environment. In vivo tests showed that this wound dressing is fully degradable and facilitates healing.
Several in vitro studies showed electrospun fiber of collagen and synthetic polymer gave better cell adhesion and proliferation compared to synthetic polymer alone. Rho et al (2006) using electrospun collagen scaffold on full thickness wound on Sprague-Dawley showed faster early stage healing (up to 7 days) compared to wound without scaffold. However, no significant difference in the wound closure was found between wound with and without collagen scaffold during later stage of the study with both groups demonstrating complete epithelialization after 4 weeks. Due to rapid degradation of collagen scaffold, it may not contribute to later stage healing of the wound. A synthetic polymer with longer degradation rate may better facilitate wound healing throughout the duration of the healing process.
The use of electrospun fibers as skin graft is not restricted to just long strand nanofibers membrane. Short strand nanofibers made from electrospinning offer several advantages over its continuous form especially when the nanofibers are used with a gel matrix. The most obvious would be the uniform distribution of short strand nanofibers which would be more difficult with 2D nanofibrous membranes. Since nanofibers have been shown to be beneficial in the adhesion and proliferation of cells, John et al (2023) used short strands electrospun poly(glycolide-co-lactide) (PGLA 90:10)/gelatin and poly-p-dioxanone (PDO)/gelatin fibers in a gelatin matrix for the construction of diabetic wound healing scaffold. To form macro-pores in the matrix, a 3D-printed alginate meshes was used as a sacrificial template and this was immersed in the gel containing the short strand fibers before freeze drying. Cross-linking was then carried out using glutaraldehyde (GA) vapor to stabilize the scaffold followed by the removal of the sacrificial template using ethylenediaminetetraacetic acid (EDTA) to create macro channels. The presence of the macro channels would facilitate cell migration and hence wound healing. To further accelerate wound healing, John et al (2023) injected W379 peptide into the gel scaffold. The 3D gel scaffold with macro channels and W379 peptide was found to greatly enhance vascularized granulation and re-epithelialization in TALLYHO type 2 diabetic mice model.
In a study by Han et al (2007) using Athymic nude mice full-thickness skin wound model, poly(3-hydroxybutyrate-co-3-hydroxyvalerate) (PHB) nanofiber scaffold showed better healing than PHB/collagen. The best result comes from PHB nanofiber scaffold seeded with cocultured (3 - 5 days) dermal sheath (DS) cells and epithelial outer root sheath (ORS) cells.
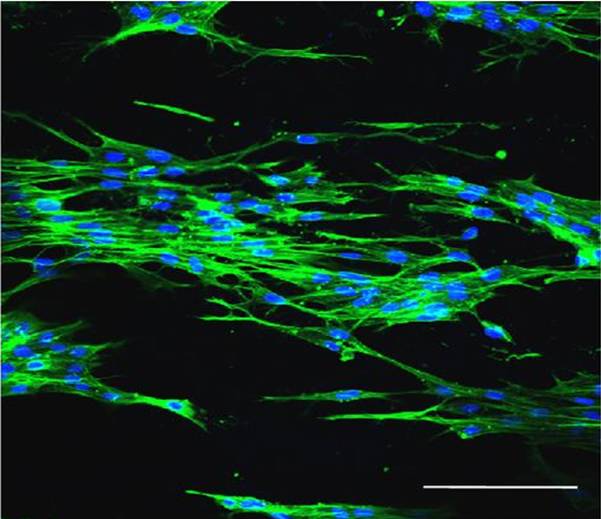
Scaffold seeded with cells may accelerate healing as they are able to immediately participate in the repair and restoration of the injury site. Different cell sources and cell types have been tested in vivo with scaffold and the results have been better healing than scaffold without cells. Apart from dermal sheath (DS) cells and epithelial outer root sheath (ORS) cells [Han et al 2007], other cells such as keratinocytes, fibroblasts and stem cells have been used. Duan et al (2013) seeded human keratinocyte cell line (HaCaT) (Seeding density of 1 x 106 cells per cm2 and cultured for 7 days) and cultured keratinocyte (KC) (Seeding density of 1 x 106 cells per cm2 and cultured for 7 days) from donors onto modified gelatin and poly¬caprolactone (GT/PCL) electrospun membrane and faster healing was observed in a full thickness BALB/c nude mice wound model. Immunofluorescence staining of anti¬human HLA-ABC on the epithelial layers showed the presence of the donor cells thus providing evidence that the cells have contributed to the healing process. Bonvallet et al (2015) showed that electrospun a 70% collagen I and 30% poly(ε-aprolactone) nanofibers membrane perforated with 160 µm diameter holes and seeded with F344 fibroblasts (35,000 cells on a 15 mm diameter scaffold) performed better than acellular membrane using a syngeneic Fischer 344 (F344) rat full-thickness skin defect model. The presence of the through pores also allowed the cells to migrate into the depth of the scaffold. F344 fibroblasts were isolated from the rat skin to avoid rejection of the implanted cells. In vitro study showed that extracellular matrix (ECM) was found to be deposited in the 160 µm diameter holes with maximum pore filling by 14 days. Comparing the performance of the scaffolds where cells have been cultured for 1 day and 4 days, the scaffold with longer cell cultivation prior to implantation showed better recovery. This could be due to a combination of higher cell density and presence of more ECM on the scaffold.
To reduce the need for cells from donor site, stem cells is an attractive alternative as it can be obtained from various sources such as bone marrow. The stem cells may potentially differentiate into the desired lineage at the wound site. Interestingly, Millán-Rivero et al (2019) found that mesenchymal stem cells (MSCs) injected at the wound periphery together with MSCs cellularized silk fibroin (SF) electrospun scaffold gave a better outcome compared to SF scaffold alone and MSCs cellularized scaffold in their hairless SKH1 mice full thickness wound model. The combination treatment gave rise to high quality, well vascularized granulation tissue, enhanced re-epithelialization of the wound and attenuated the formation of fibrotic scar tissue. It is possible that there is a synergistic effect when MSCs are injected at the wound periphery together with MSCs cellularized silk fibroin (SF) electrospun scaffold. This combination allows a higher number of MSCs to be introduced to the wound site. Further investigation showed that the extra MSCs did not migrate to other organs and there are no abnormal proliferation or formed teratomas and/or tumors in distant organs. Adipose stem cells (ASC), when seeded into electrospun scaffolds have also been shown to accelerate wound healing. Liu et al (2023) constructed a scaffold with electrospun polycaprolactone (PCL) nanofibrous yarns forming square grids as a base with a layer of randomly oriented electrospun gelatin nanofibers over it. A rat full-thickness skin defect of 13 mm in diameter was used to investigate wound healing potential of the electrospun scaffold with and without ASC seeded. On day 14, the group with ASC seeded on the electrospun scaffold showed the best results with better degree of epithelialization. Hair follicles and sebaceous glands were observed hence showing its potential for wound healing application.
A more readily available source of cell that does not require days of in-vitro expansion is bone marrow derived mesenchymal stem cells (MSC). While most approach to implanting cell-loaded scaffold involves in vitro culturing and expansion of the cells on the scaffold for several days, a better clinical approach is to seed the cells on the scaffold prior to implantation. This eliminates the risk of any contamination during culture or in transportation. Ma et al (2011) seeded MSC onto electrospun collagen-blended poly (D,L)-lactic-co-glycolic acid (PLGA) and allow the cells to adhere on the scaffold for 30 min before washing away any unattached cells and implanting the scaffold. Scaffold seeded with high density of MSC (about 3 x 106 per 10 mm x 10 mm scaffold) and implanted on Sprague Dawley rats full thickness wound showed complete wound closure by day 10 which was 8 days earlier than control group without any scaffold. Intact epithelium with hair follicles and sebaceous glands and normal collagen structure was observed which was not observable in other groups with scaffold only and scaffold with low density of MSC seeding (3 x 103 per 10 mm x 10 mm scaffold) [Ma et al 2011].
For commercial utilization of electrospun scaffold, it is important to investigate the rate of healing with commercial alternatives. Comparing healing of full thickness wound between using electrospun poly (D,L)-lactic-co-glycolic acid (PLGA) /collagen scaffold and gauze or commercial dressing on a Sprague Dawley rats demonstrated almost complete healing of wound at 3 weeks while wound treated with gauze or commercial dressing showed incomplete re-epithelialization and inflammatory cell infiltration [Liu et al 2010].
Most in vivo testing are carried out on rodents and although the results are encouraging, their skin architecture and healing is very different from human. A closer model to use is a pig model and electrospun sterilized hyaluronic acid scaffold has been compared against adhesive bandage, solid sterilized hyaluronic acid, gauge with Vaseline and an antibiotic dressing in a full thickness wound model. The results showed better healing with electrospun sterilized hyaluronic acid against all others with complete epithelialization, good wound color and wound cover after 12 days [Uppal et al 2011].
Electrospun membranes with anti-bacterial properties have been produced and this has been tested in wound healing applications. A study by Li et al (2013) showed that electrospun poly(vinyl alcohol) (PVA) and chitosan oligosaccharides (COS) with silver nanoparticles (AgNPs) scaffold applied to Sprague Dawley rats full thickness wound demonstrated complete re-epithelialization on day 14. PVA/COS/AgNPs showed better healing compared to commercial woundplast, gauze and PVA/COS/AgNO3 nanofiber with the latter two treatments showing ulcerated surfaces and formation of granulation tissue after seven days [Li et al 2013]. It is likely that slower release of Ag ions from AgNPs facilitates wound healing through longer protection duration compared to AgNO3.
Liu et al (2023) constructed a multi-layered composite structure with electrospun polycaprolactone (PCL) fibers and sprayed alginate hydrogel powder by alternating electrospinning and spraying. The multi-layered structure was wetted and cross-linked so that the alginate formed a stable matrix with the PCL fibrous layers to form a fiber hydrogel interpenetrated network (FHIPN). Amino-terminated hyperbranched polyamide (ATHBP) with antibacterial properties was added to the FHIPN by dipping the FHIPN in ATHBP solution to form functionalized fiber-hydrogel interpenetrating network (FFHIPN). In vivo study using mouse full-thickness wound defect model showed significantly better wound healing using FHIPN and FFHIPN compared to the negative control using Gauze. In particular, histological tests showed the appearance of hair follicle structures on FHIPN and FFHIPN treated mice by day 7 while a small amount of hair follicle appeared in the wounds treated with Gauze on day 14. At day 14 there was almost complete wound closure for FFHIPN treated wounds. To determine the efficacy of FFHIPN over FHIPN, P. aeruginosa was inoculated locally at the skin defects. On day 3, a significant amount of the bacteria was found on the wounds treated with FHIP while the amount of bacteria treated with FFHIPN was significantly less. On day 14, wound closure was significantly faster in the FFHIPN group compared to the FHIP group which highlights the beneficial effect of having antimicrobial skin substitute to reduce infection and promote healing.
Electrospun responsive fibers have been constructed to control the release of Ag+. Zhao et al (2023) electrospun fibers made of polycaprolactone (PCL), silver nanoparticles (AgNPs) and black phosphorus (BP), with PCL being the carrier matrix, AgNPs with antibacterial functionality and photothermal BP. When the PCL/AgNPs/BP fibers were irradiated with 808 nm near infra-red (NIR) laser at 0.8 W cm-2, the temperature was able to increase to about 41 °C in less than 2 min. With the increase in the temperature, the release rate of drugs is expected to increase concurrently. In the absence of NIR, the release of AgNPs was about 62% at 14 days. With application of NIR irradiation and an expected temperature of 41 °C, the release rate increased to 89%. The released silver nanoparticles and silver ions were found to be effective in inhibiting E. colidrugged resistant MRSA bacteria. In particular with the application of NIR irradiation, the increased release of Ag+ disrupted bacterial biofilm and the local hyperthermia induced by the BP increases bacterial sensitivity which further inhibits the bacteria. With an optimized ratio of PCL/AgNPs/BP, the composite fibers were able to show complete wound healing in a rat model at day 15 while the control group was unhealed. This demonstrated better bacterial disinfection with the AgNPs and BP.
Other capabilities have been added to electrospun fibers on top of antibacterial properties. Nejaddehbashi (2023) used electrospinning to construct a double-layered nanofibrous mat with one layer containing silver sulfadiazine (SSD) for antibacterial properties and polycaprolactone (PCL) and the other layer made of collagen and PCL. The resultant mat was immersed in grape seed extract (GSE) solution (2% Wt) overnight to impart antioxidant properties to the electrospun mat. The mat was able to inhibit gram-positive and -negative bacteria. Using a rat diabetic wound model, the double-layered electrospun mat with GSE showed fully repaired skin on day 14 while the pure mat group was not completely healed. This demonstrated the positive impact of having both SSD and GSE in the electrospun wound dressing.
Gao et al (2023) used electrospinning to produce core-shell fibers with silver nitrate and polyvinylpyrrolidone (PVP) as the shell and polycaprolactone (PCL), chitosan and Panax notoginseng (PN) as the core. The multifunctional core-shell fibers used silver nitrate for its antibacterial property, chitosan (CS) for hygroscopic property and PN which is a traditional Chinese medicine with anti-inflammation property. Having silver nitrate in the shell enables rapid inhibition of any bacteria in the wound area while the slower release of PN encourages wound healing. In vivo study using Sprague Dawley mice showed that the wound area was reduced to just 1.52% at day 9 while the control using gauze was 16.3%. At day 12, the wound covered by the multifunctional fibers membrane had mostly healed while the control still had a wound area of 10.8%.
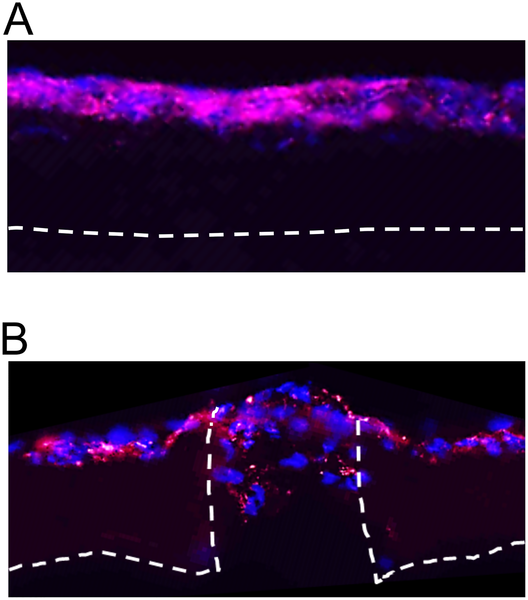
Most investigations into the use of electrospun membrane for wound healing are based on randomly oriented nanofibers. Small pore size due to the overlapping randomly oriented nanofibers of electrospun membrane typically limits infiltration of cells. There may be concerns about the lack of cell infiltration into the scaffold which may hinder the healing process in particular larger wound size. Moreover, infiltration of endothelial cells is essential for vascularization of the scaffold to maintain cellular activity. Scaffolds with aligned nanofibers have been found to facilitate endothelial cell infiltration into it especially with heparin coating on the nanofibers. Kurpinski et al (2010) demonstrated this effect with electrospun poly(L-lactide) (PLLA) nanofibers scaffold on a rat full thickness wound model. However, there were no obvious differences in epidermal growth into wound area and macroscopic healing between randomly oriented or aligned fibers scaffold. A known method of increasing pore size of electrospun membrane is to increase its fiber diameter to several microns. However, this would negate the benefit of constructing fibers in the nanometer scale using electrospinning. A layered structure with micro-diameter fiber at the base and nanofiber at the top may combine the beneficial characteristic of both dimensions. Chen et al used higher solution concentration to electrospin poly(DL-lactide) base layer with fiber diameter of about 2.5 µm. The upper layer comprises of aligned chitosan fibers with diameter of about 243 nm. Full thickness rat wound model showed good restoration of the epidermis and dermis layer with obvious separation using the layered composite with regeneration of keratinocytes and fibroblasts respectively. Cell infiltration up to 32 µm depth was observed which facilitates skin tissue remodeling. This contrasts with the lack of distinct separation of the skin layers where sterilize gauze, PDLLA films and chitosan fibers were used [Chen et al 2014].
Yuan et al (2024) used a gas foaming technique to construct a 3D scaffold made of woven electrospun aligned nanofiber strips. A rotating drum collector was used to form a membrane made of aligned electrospun polycaprolactone (PCL). The membrane was then cut into strips of PCL fibers after soaking in liquid nitrogen for a few minutes. The fiber strips were then woven into criss-crossing "bamboo basket" scaffolds using weaving technology. The woven scaffold edges were subsequently heat sealed to stabilise its structure. Foaming using sodium borohydride solution was carried out for 10 min followed by washing and freeze-drying. With the expansion by foaming, the average pore size of the scaffold was 200 µm and the thickness of the scaffold was 1.79 mm. Using an in vivo mouse diabetic skin wound model, the orthogonally woven 3D PCL nanofiber scaffolds showed faster wound closure compared to PELNAC material (commercial), and Lando material (commercial). The orthogonally aligned nanofibers may have facilitated the migration of fibroblasts and keratinocytes from surrounding host tissue. Greater monocyte and neutrophil infiltration was also observed in the orthogonally woven 3D nanofiber scaffolds and accelerated granulation tissue formation. Higher expression of pro-regenerative inflammatory markers were also detected from day 3 onwards with complete re-epithelialization on day 14.
Further investigation is required to determine the significance of better cell infiltration into scaffold for wound healing especially for larger wound area in larger animals.
Methods to construct 3D scaffolds from electrospun fibers generally exhibit higher pore size and porosity. Liu et al (2025) used a homogeniser to obtain a suspension of electrospun gelatin (Gel)/polylactic acid (PLA)/magnesium oxide nanoparticles (MgO) nanofibers. The suspension of nanofibers was subsequently poured into a mold and freeze dried to form a 3D nanofibers scaffold. Crosslinking was carried out at 180°C for 2h to form the final aerogel scaffolds. Comparing the Gel/PLA scaffolds with and without MgO nanoparticles, the scaffold containing MgO had a porosity of 80% which was significantly greater than the scaffold without MgO at 68%. The aerogel Gel/PLA/MgO scaffold tested on a rat full thickness diabetic wound model showed excellent healing with recovery of epidermal tissue and regeneration of hair follicles and hair by day 21.
3D printing and electrospinning has been used in a multi-layered approach to the construction of a skin scaffold. Having structural variation in the scaffold may better meet the functional requirements across the skin thickness. In the construction of a skin graft, Pajooh et al (2024) 3D printed dextran-vascular endothelial growth factor (Dex-VEGF) as the top layer to stimulate angiogenesis and cell migration and functions as the epidermis to retain moisture. The bottom layer was made of electrospun gelatin-keratin (Gel-Kr) nanofibers to induce cell attachment and function as the dermis due to its structural similarity to ECM. In vivo study using Male Wistar rats full-thickness dorsal wound showed that wound healing was the fastest with bilayered 3D printed (3DP) Dex-VEGF/nanofibers membrane scaffold at 97.1%. This is better than the 3DP Dex-VEGF scaffold with 87.16% wound closure and 68.03 % for the 3DP group and 56.63 % for the control group. The healing process for 3DP Dex-VEGF/nanofibers membrane scaffold and 3DP Dex-VEGF were in the late stages with lowest number of inflammatory cells and neovascularization areas.
Electrospun membrane has shown to be a good candidate for treating full thickness wound. While researchers have mostly favored using or blending natural protein with synthetic polymer in the construction of the scaffold, the right choice of synthetic polymer has been shown to perform better than scaffold with natural polymer blended. Studies so far has shown that scaffold with cells seeded perform better than just purely scaffold. It has also been demonstrated that rapid adhesion of MSCs on scaffold in 30 min prior to implantation was able to give much better healing than pure scaffold thus providing a clinically advantages method for cell-scaffold implants. The use of electrospun scaffold has also been shown to be superior to some commercially available treatment methods. Using a porcine model instead of a rodent model also showed good treatment outcome using electrospun scaffold. Although in vivo studies on electrospun scaffold has been focused on material and with or without cell seeding, other aspects of the scaffold such as fiber orientation and pore size may yield interesting outcomes.
Published date: 01 October 2014
Last updated: 28 April 2026
▼ Reference
-
Bonvallet PP, Schultz MJ, Mitchell EH, Bain JL, Culpepper BK, Thomas SJ, Bellis S L. Microporous Dermal-Mimetic Electrospun Scaffolds Pre-Seeded with Fibroblasts Promote Tissue Regeneration in Full-Thickness Skin Wounds. PLoS ONE 2015; 10(3): e0122359. doi:10.1371/journal.pone.0122359.
Open Access
-
Chen S H, Chang Y, Lee K R, Lai J Y. A three-dimensional dual-layer nano/microfibrous structure of electrospun chitosan/poly(D,L-lactide) membrane for the improvement of cytocompatibility. Journal of Membrane Science 2014; 450: 224.
-
Duan H, Feng B, Guo X, Wang J, Zhao L, Zhou G, Lu W, Cao Y, Zhang W J. Engineering of epidermis skin grafts using electrospun nanofibrous gelatin/polycaprolactone membranes. International Journal of Nanomedicine 2013; 8: 2077.
Open Access
-
Gao Z, Liu S, Li S, Shao X, Zhang P, Yao Q. Fabrication and Properties of the Multifunctional Rapid Wound Healing Panax notoginseng@Ag Electrospun Fiber Membrane. Molecules. 2023; 28(7):2972.
Open Access
-
Han I, Shim K J, Kim J Y, Im S U, Sung Y K, Kim M, Kang I K, Kim J C. Effect of poly(3-hydroxybutyrate-co-3-hydroxyvalerate) nanofiber matrices cocultured with hair follicular epithelial and dermal cells for biological wound dressing. Artif Organs 2007; 31: 801.
-
John J V, Sharma N S, Tang G, Luo Z, Su Y, Weihs S, Shahriar S M S, Wang G, McCarthy A, Dyke J, Zhang Y S, Khademhosseini A, Xie J. Nanofiber Aerogels with Precision Macrochannels and LL-37-Mimic Peptides Synergistically Promote Diabetic Wound Healing. Advanced Functional Materials 2023; 33: 2206936.
Open Access
-
Kang Y O, Yoon I S, Lee S Y, Kim D D, Lee S J, Park W H, Hudson S M. Chitosan-Coated Poly(vinyl alcohol) Nanofibers For Wound Dressings. J Biomed Mater Res Part B: Appl Biomater 2010; 92B: 568.
-
Khil M S, Cha D I, Kim H Y, Kim I S, Bhattarai N. Electrospun Nanofibrous Polyurethane Membrane as Wound Dressing. J Biomed Mater Res Part B: Appl Biomater 2003; 67B: 675.
-
Kurpinski K T, Stephenson J T, Janairo R R R, Lee H, Li S. The effect of fiber alignment and heparin coating on cell infiltration into nanofibrous PLLA scaffolds. Biomaterials 2010; 31: 3536.
-
Li C, Fu R, Yu C, Li Z, Guan H, Hu D, Zhao D, Lu L. Silver nanoparticle/chitosan oligosaccharide/poly(vinyl alcohol) nanofibers as wound dressings: a preclinical study. International Journal of Nanomedicine 2013; 8: 4131.
Open Access
-
Liu M, Chen Y, Zhang Y, Zhuang P, Wang J. Breathable functional aerogel dressings facilitate the healing of diabetic wounds. Biomedical Technology 2025; 9: 100071.
https://www.sciencedirect.com/science/article/pii/S2949723X25000030 Open Access.
-
Liu N, Zhou Z, Ning X, Zhang X, Guo Q, Guo M, Wang Y, Wu T. Enhancing the paracrine effects of adipose stem cells using nanofiber-based meshes prepared by light-welding for accelerating wound healing. Materials & Design 2023; 225: 111582.
Open Access
-
Liu S J, Kau Y C, Chou C Y, Chen J K, Wu R C, Yeh W L. Electrospun PLGA/collagen nanofibrous membrane as early-stage wound dressing. Journal of Membrane Science 2010; 355: 53.
-
Liu X, Gao L, Fu S, Zhao W, Wang F, Gao J, Li C, Wu H, Wang L. Polycaprolactone nanofiber-alginate hydrogel interpenetrated skin substitute for regulation of wound-substitute interface. Materials & Design 2023; 227: 111706.
Open Access
-
Ma K, Liao S, He L, Lu J, Ramakrishna S, Chan C K. Effects of Nanofiber/Stem Cell Composite on Wound Healing in Acute Full-Thickness Skin Wounds. Tissue Engineering Part A 2011; 17: 1413.
-
Millán-Rivero J E, Martinez C M, Romecín P A, Aznar-Cervantes S D, Carpes-Ruiz M, Cenis J L, Moraleda J M, Atucha N M, García-Bernal D. Silk fibroin scaffolds seeded with Wharton's jelly mesenchymal stem cells enhance re-epithelialization and reduce formation of scar tissue after cutaneous wound healing. Stem Cell Res Ther 2019; 10: 126.
Open Access
-
Nejaddehbashi F, Rafiee Z, Orazizadeh M, Bayati V, Hemmati A, Hashemitabar M, Makvandi P. Antibacterial and antioxidant double-layered nanofibrous mat promotes wound healing in diabetic rats. Sci Rep 2023; 13: 3166.
Open Access
-
Pajooh A M D, Tavakoli M, Al-Musawi M H, Karimi A, Salehi E, Nasiri-Harchegani S, Sharifianjazi F, Tavamaishvili K, Mehrjoo M, Najafinezhad A, Varshosaz J, Mirhaj M. Biomimetic VEGF-loaded bilayer scaffold fabricated by 3D printing and electrospinning techniques for skin regeneration. Material & Design 2024; 238: 112714.
Open Access
-
Rho K S, Jeong L, Lee G, Seo B M, Park Y J, Hong S D, Roh S, Cho J J, Park W H, Min B M. Electrospinning of collagen nanofibers: effects on the behavior of normal human keratinocytes and early-stage wound healing. Biomaterials 2006; 8: 1452.
-
Sen S, Bal T, Rajora AD. Green nanofiber mat from HLM-PVA-Pectin (Hibiscus leaves mucilage-polyvinyl alcohol-pectin) polymeric blend using electrospinning technique as a novel material in wound-healing process. Appl Nanosci 2022; 12: 237.
Open Access
-
Uppal R, Ramaswamy G N, Arnold C, Goodband R, Wang Y. Hyaluronic acid nanofiber wound dressing-production, characterization, and in vivo behavior. J Biomed Mater Res Part B: Appl Biomater 2011; 97B: 20.
-
Yuan J, Sun B, Ma W, Cai C, Huang Z, Zhou P, Yi L, Liu L, Chen S. Orthogonally woven 3D nanofiber scaffolds promote rapid soft tissue regeneration by enhancing bidirectional cell migration. Bioactive Materials 2024; 39: 582.
https://www.sciencedirect.com/science/article/pii/S2452199X24001567 Open Access.
-
Zhao Y, Liu Y, Tian C, Liu ZZ, Wu K, Zhang C, 7an X. Construction of antibacterial photothermal PCL/AgNPs/BP nanofibers for infected wound healing. Materials & Design 2023; 226: 111670.
Open Access
▲ Close list
 ElectrospinTech
ElectrospinTech