Bacteria and microbes are part of the eco-system and many species are used in biotechnology applications. The advantage of electrospun membrane is its high porosity and large surface area which maximises any interaction between trapped bacteria and the environment. There are a few methods in which electrospinning may be used to contain bacteria and microbe. The more commonly used technique is for the bacteria to be encapsulated within the nanofiber matrix. A less common technique is to use the electrospun membrane as a physical barrier to encapsulate the bacteria within a pocket.
The pore sizes between the fibers of electrospun membrane are generally too small for the bacteria to pass through. Lev et al (2012) showed that electrospun polyurethane nanofibers on polypropylene substrate are able to remove E. coli from water therefore demonstrating that the nanofibrous membrane provides a physical barrier to the bacteria. Jesus et al (2014) showed that electrospun scaffold of poly(L-lactide) with fiber diameter of 800 nm to more than 1 µm is able to support E. coli and S. aureus adhesion after 5 minutes of incubation. To demonstrate the use of bacteria immobilized on electrospun membrane in wastewater treatment, San et al (2014) immobilized three types of bacteria (Aeromonas eucrenophila, Clavibacter michiganensis and Pseudomonas aeruginosa) on cellulose acetate electrospun membrane. Using methylene blue as the contaminant, they showed that all three bacteria were able to achieve more than 95% removal efficiency which is comparable to removal from free bacteria. However, more works need to be done to reduce the amount of bacteria lost after subsequent washing step as the efficiency drops to below 50% after the forth cycle of washing. Abrigo et al (2015) showed that the surface chemistry of the electrospun nanofibers have a significant effect on the attachment of bacteria. Using interactions of Escherichia coli(E. coli) on surface modified fibers by plasma treatment, they found that hydrophobic 1,7-octadiene (ppOct) treated mesh resulted in higher proportion of live bacteria compared to untreated PS mesh and hydrophilic acrylic acid (ppAAc) surface treated PS mesh has significant lower E coli. To avoid loss of bacteria, another method is to encapsulate the bacteria within the fiber matrix.
Size exclusion using electrospun membrane may be used for separation of microorganisms. Suaste-Gómez et al (2022) investigated the use of electrospun membrane as biological sieve for the separation of yeast and bacteria in detecting the origin of vaginal infection. The yeasts used in the test was Candida albicans type with dimensions of 7 µm and the bacteria was Brevibacterium ravens purgense with dimensions of 1.4 µm. Electrospun polylactic acid (PLA) of thickness 66 µm, 83 µm and 128 µm were used in the study. With the thinner membranes, some yeasts were found to pass through the membrane while the 128 µm thick membrane was able to reject all the yeasts. Counterintuitively, it was reported that there was no bacteria passing through the thinnest membrane (66 µm) while the thickest membrane (128 µm) reported the most bacteria passing through. Other independent studies will be required to validate this observation.
Microbes and bacteria have also been encapsulated within fibers using electrospinning. Salalha et al (2006) tested the viability of Escherichia coli and Staphylococcus albus following electrospinning. Viability of the bacteria were found to be better with 19% for E. coli and 100% for S. albus while those of the virus were poor with less than 10% viability. These differences were attributed to the presence of cell wall for the bacteria which are better able to withstand the shear force during electrospinning. Salalha et al (2006) hypothesized that it is the rapid vaporization of the solvent during electrospinning resulted in the relatively poorer viability of E. coli compared to S. albus. By culturing E. coli in 5% glycerol, the viability of E. coli was significantly raised to 48%. The presence of glycerol in the E. coli probably protects the cell from rapid dehydration during electrospinning. Lopez-Rubio et al (2009) tested the viability of Bifidobacterium animalis subsp. lactis Bb12 when encapsulated in polyvinyl alcohol (PVA) in a core-shell setup. B animalis Bb12 was suspended in skimmed milk and encapsulated at the core of the fiber. Comparing the viability of B animalis Bb12 encapsulated in the fiber with non-encapsulated B animalis Bb12, viability of the encapsulated B animalis Bb12 was significantly higher when both were stored at 4 °C for 130 days. When stored at room temperature and - 20 °C, both showed reduced viability over time. A potential application is in the delivery of microbes and bacteria to specific organs for re-establishment of a healthy population of microbiota. Nagy et al (2014) investigated the use of water soluble polymers (polyvinyl alcohol and polyvinylpyrrolidone) for encapsulation and delivery of Lactobacillus acidophilus bacteria. The survival rate of the bacteria was found to be between 34% and 68%.
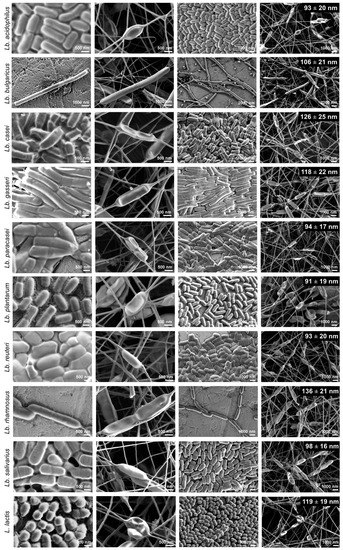
Zupancic et al (2019) studied the effects of electrospinning on the viability of ten species of lactic acid bacteria in poly(ethylene oxide) (PEO) nanofibers. While all the bacterial species are viable following encapsulation in the electrospun nanofibers, their survivability varies. Five species, Lactobacillus (Lb.) acidophilus, Lb. gasseri, Lb. reuteri, Lb. salivarius, and L. lactis showed high viability with survival rate of about 90% and Lb. delbrueckii ssp. bulgaricus was the worst with survival rate of about 50%. Lb. casei, Lb. paracasei, Lb. plantarum, and Lb. rhamnosus showed survival rate in between. In this study, hydrophilic bacteria showed lower survival rate than hydrophobic bacteria. The fact that electrospinning of PEO with the bacteria is done in aqueous media may result in greater shear force on hydrophilic bacteria compared to hydrophobic bacteria. While there are no general correlation found between bacteria size and bacteria viability, the low viability of Lb. delbrueckii ssp. Bulgaricus may be related to its size as it has by far the longest cells.
This electrospun membrane has been developed for potential treatment of bacterial vaginosis. In which case, the water soluble polymer will release its bacteria loads upon application.
Stojanov et al (2024) investigated the potential use of electrospun fibers as a platform for delivery of vaginal lactobacilli modified with fluorescent (mCherry and GFP) and luminescent (NanoLuc luciferase) proteins. Modified lactobacilli (
Lactobacillus crispatus,
Lactobacillus gasseri and
Lactobacillus jensenii) were loaded into polyethylene oxide (PEO) solution for electrospinning into fibers. The release of the bacteria can be monitored through the fluorescence signal and it was shown that about 20 - 60% of the bacteria were released in the first 10 min of incubation and all the bacteria were released in 30 - 45 min. Viability of the fluorescent protein-labelled bacteria after electrospinning to form fibers differed with the most decreased shown by
L. crispatus and less decreased by
L. gasseri and L. jensenii. Of these three bacteria,
L. gasseri and L. jensenii showed high hydrophobicity at 95.8% and 87.3% respectively, as determined by extracting them with n-hexadecan. L. crispatus showed much lower hydrophobicity at 21.4%. The effect of bacteria hydrophobicity on its viability is consistent with the results from
Zupancic et al (2019) where more hydrophilic bacteria may suffer from greater shear force when electrospinning in an aqueous media resulting in reduced viability.
Another targeted organ for re-introduction of beneficial microbes is the gastro-intestinal tract.
For utilization of microbes in biotechnology, encapsulation of microbes in fibers will ensure retention of the microbes in the treatment area. However, this is not sufficient as the microbe must be able to interact with the environment despite been surrounded by the matrix material. Tong et al (2013) tested the functionality of E. coli bacteria encapsulated in electrospun silica/polyvinyl alcohol (PVA) fiber with free E. coli. From the degradation of atrazine, they found that the encapsulation did not significantly impede the activity of E. coli. Further the formation of silica/PVA using silica precursor results in a water-insoluble fiber matrix. Rendering a water-soluble material insoluble is important for practical application of microbe-encapsulated fibers in cases where the material is not designed to release the microbes. Liu et al (2009) used water-soluble Pluronic F127 dimethacrylate (FDMA) and polyethylene oxide (PEO) to mix with Pseudomonas fluorescens, Zymomonas mobilis, and E. coli for electrospinning. To cross-link the fibers to render the fibers water insoluble, glycerol was used as the cross-linking medium as it has low toxicity to microorganism. A redox system consisting of ammonium persulfate (APS), ascorbic acid (AsA), and ferrous sulfate was added to the solution of glycerol and water for cross-linking of the fibers. The fibrous structure was retained after removal of PEO. Z mobilis was found to remain viable after storage at 4 °C for up to 7 days although the percentage of bacteria that remained viable was reduced to 23%. The metabolic activity of Z mobilis was not affected by the electrospinning process.
Encapsulation of microbes in electrospun nanofibers is also useful in agricultural applications. Damasceno et al (2013) used electrospun polyvinyl alcohol (PVA) nanofibers to encapsulate rhizobia, an economically interesting bacterium found in legumes. The PVA matrix potentially protects the rhizobia from environmental stress such as temperature and dehydration. Comparing the viability of rhizobia encapsulated in PVA nanofiber and the negative control (unprotected rhizobia), significantly more rhizobia remains viability after 48 h of storage for those encapsulated in PVA nanofiber. Study was also carried out to determine whether the encapsulation of the bacteria in PVA nanofiber has a negative impact on the number of nodules present in soybean when applied. There is no significant difference in the number of nodules formed between rhizobia encapsulated in PVA nanofiber and the positive control (unprotected rhizobia) over a 30 days period. Therefore, encapsulation of rhizobia in PVA nanofibers is a viable method for storage and delivery of the bacteria.
Electrospun membrane may be used indirectly for power generation in the form of a supporting and conductive substrate for inoculation of electrochemically active bacteria. Guzman et al (2017) explored such a bioelectrochemical systems (BESs) using electrospun polyacrylonitrile(PAN)-derived carbon nanofiber (CNF) coated with the conductive polymer poly(3,4-ethylenedioxythiophene) (PEDOT) and inoculated with Geobacter sulfurreducens in a bioreactor. Using PEDOT coated carbon nanofiber and inoculated with Geobacter sulfurreducens, a current of 10.66 A m-2 was recorded. However, this was lower than PEDOT coated carbon cloth with the bacteria which gave a current of 15.22 A m-2. In terms of output normalized by mass, high porosity of carbon nanofiber outperforms carbon cloth.
Amen et al (2022) constructed the microbial fuel cells (MFC) with electrospun membrane as the anode material. High specific area and porosities of electrospun carbon fiber membrane should allow high electron transfer capabilities and potentially addresses the issues of low power and current densities in most MFCs. The network of fibers may also encourage the formation of biofilm on the anode by the bacteria.
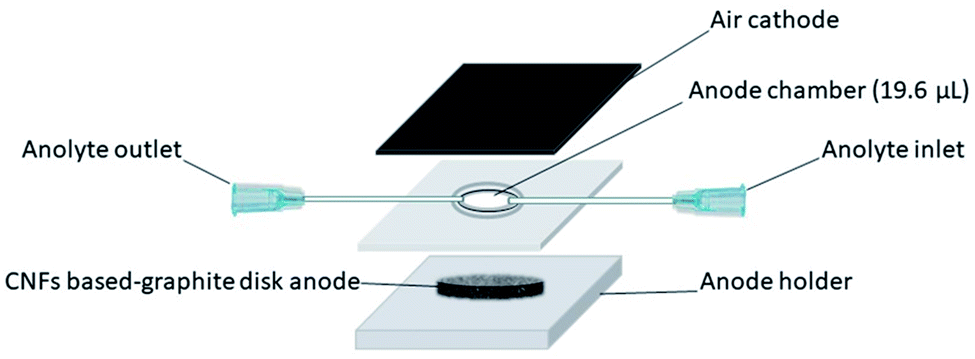
In their setup, the carbon nanofibers were prepared by electrospinning and carbonizing polyacrylonitrile (PAN). Escherichia coli 0157 NCCP-14541 was used as the electroactive bacteria in the anode chamber. As MFC uses living bacteria to generate electricity, the power generation varies over incubation days. For their optimum setup, the power generated after the first 24 h was 2.24 Wm-2 at 7.89 Am-2. The maximum power output of 8.1 Wm-2 at 19.34 Am-2 was recorded after 6 days of operation while the maximum current output of 44.9 Am-2 was recorded after 5 days of running time. However, the power and current was not sustained throughout the 8 days of measurement. The optimum thickness of the membrane was found to be 10 µm with less current production at 48 h for 5 µm and 10 µm thickness membrane. This could be due to the degree of penetration of the bacteria and biofilm into the anode membrane. For a thinner membrane, the density of carbon nanofibers was lower hence the maximum current is not realized. When the membrane is too thick, the inability of the bacteria to penetrate into the full thickness increases the distance which the electrons have to travel. Wu et al (2023) prepared an electrospun Mo-doped carbon nanofibers scaffold as the anode in a H-type double-chamber microbial fuel cells (MFCs) inoculated with Shewanella putrefaciens CN32 (S. putrefaciens CN32) cell suspension. The resultant MFC showed an output power density of about 1287.38 mW·m-2 with a peak current density of 306.1 µAcm-2. S. putrefaciens CN32) formed a good biofilm with large number of bacteria on the surface of Mo2C nanofiber scaffold compared to scarce bacteria count on the surface of pure carbon nanofiber anode. This demonstrated the improved biocompatibility of Mo2C which led to better MFC performance. At the third discharge cycle, the Mo2C nanofiber anode produced an output current density of 0.20 mA cm-2 compared to 0.14 mA cm-2 from the carbon nanofiber anode. Further, the rough surface of Mo2C nanofibers scaffold increases the number of active sites and encourages the adsorption of extracellular bacterial proteins.
Under the right solution condition, microbes and other bioorganisms may be directly electrospun into fibers. Sugimoto et al (2019) showed that it was possible to electrospin a solution of M13 bacteriophage (phage) nano- and microfibers without additional carrier polymers. The prepared phage solution has a concentration 40-80 mg/ml in 12.5 and 25 mM Tris-buffered saline (TBS), and deionized water (DIW). The M13 phage is a long rod shape bacterial virus. Due to its anisotropic shape, it can form lyotropic liquid crystalline structures and this can form a viscous suspension in various solvents. The resulting phage fibers showed high crystallinity with phage fibrils oriented in the direction of the fiber axis. The average diameter of the phage fibers were about 150 nm with large diameter distribution probably due to the inhomogeneity of the suspension. Due to the intrinsic charged surface of the phage, Sugimoto et al (2019) was able to demonstrate the electrostatic stimulated, mechanical response of the phage fibers which gives it the potential use as electric resonator and electrostatic-stimulus responsive actuator.
Another aspect of biotechnology is to use immobilized protein for biochips and biosensors. High surface area of electrospun nanofibers made it very attractive for such application. However, it is vital that the immobilized protein is able to maintain its activity for its intended purpose. Morita et al (2017) showed that the diameter of polyurethane fibers affect the activity of adsorbed protein. Using model protein, horseradish peroxidase (HRP), with thicker fibers (diameter 950 nm), there is a decrease in activity due to large conformational changes. However, on smaller diameter fibers (diameter 480 nm), the protein was able to retain a close-to-natural shape which translate to higher activity. Their study showed that the amount of HRP adsorbed were independent of fiber diameter therefore the reduction of activity was not due to amount of adsorbed HRP. Investigating the secondary structure of adsorbed HRP on the fibers showed significantly less α-helix and β-helix percentages on the HRP adsorbed on thicker fibers compared to natural state HRP. However, the percentage of α-helix and β-helix on smaller diameter fibers were similar to natural state HRP indicating almost no structural change in the protein.
In cases where the suitable material for encapsulation of microbes is not suitable for electrospinning into fibers, electrospinning may be used instead as a carrier substrate. Stoyanova et al (2025) used simultaneous electrospinning of poly(3-hydroxybutyrate) (PHB) and electrospraying of Chitooligosaccharide (COS) containing microorganisms Bacillus subtilis to create a composite membrane as an eco-friendly, biocontrol agent to combat plant diseases and improving soil health. The B. subtilis in the composite membrane remained viable after storing in a desiccator at 4 °C for 90 days. The efficacy of the composite membrane was tested against fungal phytopathogens belonged to the genera Alternaria and Fusarium. For composite membranes containing B. subtilis, there was a zone of inhibition around it after 7 days which demonstrates suppression of pathogenic fungal growth while the control composite membrane without B. subtilis remained unchanged.
Electrospun fibers may be used to create a favorable environment to increase the performance and activity of enzymes. Most lipases perform catalytic action at hydrophilic-hydrophobic interfaces. Zhang et al (2023) demonstrated the use of electrospun fibers to create numerous hydrophilic-hydrophobic interfaces to achieve high catalytic efficiency. Two construction strategies were tested using electrospun fibers. The first method was to electrospin hydrophobic polyurethane (PU) and hydrophilic polyacrylic acid (PAA) fibers using separate nozzles but simultaneously on a collector. This will result in a nonwoven, evenly distributed mix of hydrophobic and hydrophilic fibers (PUf/PAAf). The second method was to blend hydrophobic ethyl cellulose (EC) and hydrophilic PAA polymers into a single solution followed by electrospinning into a EC/PAA fibrous membrane. The constructed membranes were subsequently heat treated for thermal cross-linking. Lipase was introduced into the membranes by soaking them in a lipase solution. The membranes would swell as it absorbed the water and took in the lipase. Hybrid membrane reactors (HMRs) were assembled by stacking the membranes and placing it in p-NPP isooctane solution. Both PUf/PAAf membrane and EC/PAA blended membrane performed much better than conventional macroscopic "oil-up/water-down" systems and emulsion systems. PUf/PAAf membrane and EC/PAA blended membrane showed a respective 2.56-fold and 2.12-fold improvement in specific activity of lipases over macroscopic "oil-up/water-down" systems and respective 1.93-fold and 2.01-fold over emulsion systems.
In 2020, the Covid-19 pandemic has raised the awareness of the importance of rapid development in vaccines and its transportation. Electrospinning has been shown to be a suitable technique for loading drugs and biologics into a nanofibrous protective polymer matrix. Dowlath et al (2021) demonstrated the potential use of electrospinning to encapsulate virus-like particle (VLP) vaccine. This VLP is derived from rabbit hemorrhagic disease virus modified to carry the MHC-I gp100 tumor-associated antigen epitope. The VLP was blended into a polyvinylpyrrolidone (PVP) solution followed by electrospinning into fibers. The electrospinning process dries the VLP while the polymer matrix protects the VLP from environmental damage. Prior to vaccination, the VLP loaded nanofiber samples were dissolved in PBS and added with CpG oligonucleotide as an adjuvant. In vivo tests were carried out on mice by subcutaneous administration to the left flank. VLP released by electrospun nanofibers was shown to induce comparable antibody titers to that of the VLP delivered in PBS. This shows that electrospinning can be used to process VLP into a dry formulation nanofiber for reconstituting prior to delivery and maintaining its immunogenicity.
In most environments, microorganisms do not exist as a monoculture. Instead, a community of microorganisms would form a biofilm on surfaces. On human bodies, such biofilm may cause infections that are resistant to the host immune system and even to antibiotics. To enhance the understanding of biofilms in human body systems, researchers are trying to develop biofilms in vitro. Biagini et al (2022) tested the adhesion of human gut microbiota microorganisms on electrospun substrates to develop a 3D in vitro model of the human gut microbiota. Gelatin and polycaprolactone (PCL) was selected for this purpose with gelatin being a natural, hydrophilic material and PCL, a synthetic and hydrophobic material. Mucin, a protein produced by epithelial tissues, was coated on half the samples of electrospun gelatin and PCL scaffolds. Interestingly, on the 24 h and 48 h time points, electrospun uncoated PCL scaffold and mucin-coated PCL scaffolds performed better than gelatin scaffolds. However, at 72 h and 7 days, uncoated gelatin scaffolds outperform all other scaffolds. Between gelatin and PCL, gelatin could be digested by microorganisms and their degradation over time may have helped to support their proliferation. PCL is more resistant to degradation and its degraded products may not support proliferation of the microorganisms.
Formation of biofilm on electrospun membranes may also find use in water treatment. Biofilm utilizes micro-organisms to consume or convert organic pollutants dissolved in the water to less harmful compounds. Zhou et al (2024) demonstrated the potential of electrospun polyacrylonitrile (PAN) membrane and PAN/polyethylene oxide (PEO) membrane as substrate and carrier for establishing and maintaining biofilm for water treatment. The experimental membranes were hung in wastewater to determine the biofilm growth. Three types of materials, electrospun PAN membrane, electrospun PAN/PEO membrane and nonwoven fabric were used in the study. While all three materials showed increasing biofilm formation, PAN/PEO nanofibers membrane showed the fastest biofilm growth and the nonwoven fabric the least. On day 3, the growth of biofilm for PAN/PEO nanofibers membrane, PAN nanofibers membrane and nonwoven fabric was 90.36%, 82.04% and 27.32% respectively. The much better performance of electrospun nanofibers membrane over nonwoven fabric may be attributed to the greater porosity and higher water absorption of the nanofibers membrane which encourages faster adhesion of microbes and more adsorption of compounds in the waste water to feed the microbes. The addition of water soluble PEO may increase water absorption of the PAN/PEO membrane which further encourages biofilm growth. Zhuo et al (2024a) examined the use of biofilms on electrospun polymethyl methacrylate (PMMA) membrane in waste water treatment by testing its ammonia (NH3) adsorption capacity, chemical oxygen demand (COD) and ammonia nitrogen concentration removal. At a relative pressure of 1.0, the NH3 adsorption capacity of 21.3685 cm3/g was much higher than that of nonwoven fabric at 21.3685 cm3/g. The ability of the membrane with biofilm to degrade organic matter in wastewater may be measured by the chemical oxygen demand (COD). With the PMMA nanofiber biofilm membrane, the COD concentration reduction showed a removal rate of 80.97% in the first two days and continued to increase while the nonwoven fabric biofilm showed a removal rate of 76.59% but with no further increase. Removal of ammonia nitrogen was also better in PMMA nanofiber biofilm membrane with a removal rate of 18.37% while the ammonia nitrogen concentration increased in nonwoven fabric groups. The better performance of PMMA nanofiber biofilm membrane may be attributed to a larger number of micro-organisms attached to it compared to non-woven fabric biofilm.
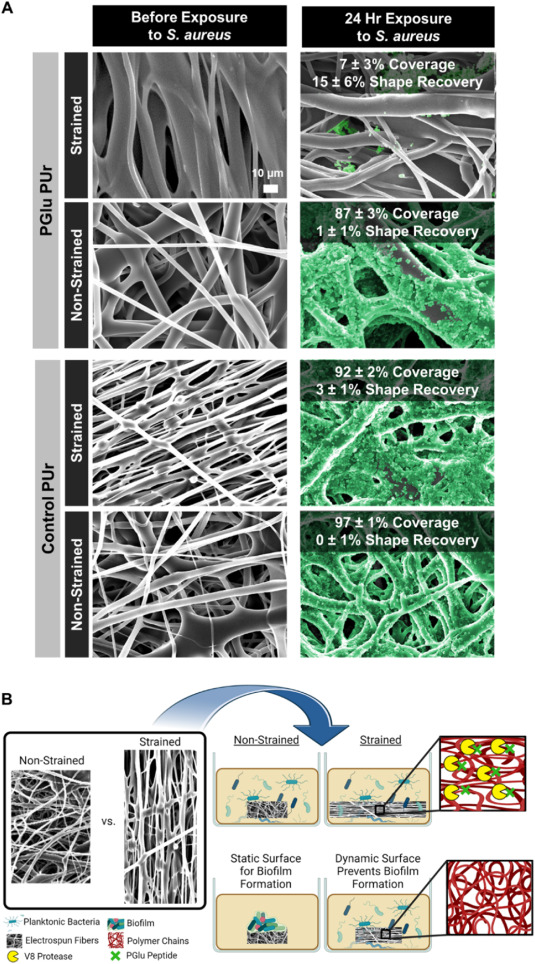
In some applications, it is desirable to discourage biofilm formation. An interesting concept in preventing bacteria from forming a biofilm has been tested for a bacteria-responsive wound dressing. Gunderson et al (2024) electrospun a polyglutamic acid-containing polyurethane (PGlu PUr) membrane and induced strain in the membrane by depositing on a rotating collector. The polyglutamic acid (PGlu) incorporated into the polymer is susceptible to degradation by V8 protease released by Staph. aureus. Enzymatic cleavage of PGlu in the backbone by V8 caused the release of the strain in the membrane and the subsequent recovery of the membrane disrupted bacteria adhesion and inhibited biofilm formation. The same effect was seen with cultures of Staph. aureus, Staph. epidermidis, and E. coli on the membrane. On controlled, non-strained PGlu PUr membrane, no recovery of the membrane was seen as biofilm formed on its surface. The inhibition by the strained PGlu PUr membrane was observed in a 24 h incubation where bacteria coverage was only about 7% while non-strained membranes were completely covered with biofilm (87 - 97%).
The ability of microbes to form biofilm on membranes has implications on the use of the membrane in various applications. Where the membrane encourages biofilm formation, it may be used in water treatment. Conversely, if the membrane discourages biofilm formation, it may be used as packaging material for food preservation. The response of microbes on electrospun membrane depends on the material of the membrane, the diameter of the fibers forming the membrane and the specific microbe. Lencova et al (2024) tested the response of different strains of Escherichia coli (ATCC 25922 and ATCC 8739) and Staphylococcus aureus (ATCC 25923 and ATCC 6538) on electrospun poly-ε-caprolactone (PCL) membranes. All tested bacteria strains biofilm formation was better on polystyrene plate compared to the electrospun membrane. With membranes of two different electrospun fiber diameters, only for strain E. coli 8739 was there significant differences in the amount of biofilm formation while the others showed no effect. However, with smaller diameter fibers (376 nm), all the bacteria strains formed a uniform biofilm layer on the surface of the membrane but with larger fiber diameters (945 nm), the bacteria formed smaller clusters inside the membrane. This is probably due to the pore size between the fibers with small diameter fibers forming smaller pore size which prevented the bacteria from migrating into the membrane.
Published date: 29 September 2015
Last updated: 12 May 2026
▼ Reference
-
Abrigo M, Kingshott P, McArthur S L. Bacterial response to different surface chemistries fabricated by plasma polymerization on electrospun nanofibers. Biointerphases 2015; 10: 04A301.
-
Amen M T, Kim H Y, Barakat N A M. Three-dimensional carbon nanofiber-based anode for high generated current and power from air-cathode micro-sized MFC. RSC Adv. 2022; 12: 15486.
Open Access
-
Biagini F, Calvigioni M, De Maria C, Magliaro C, Montemurro F, Mazzantini D, Celandroni F, Mattioli-Belmonte M, Ghelardi E, Vozzi G. Study of the Adhesion of the Human Gut Microbiota on Electrospun Structures. Bioengineering. 2022; 9(3):96.
Open Access
-
Damasceno R, Roggia I, Pereira C, Sa E. Rhizobia survival in seeds coated with polyvinyl alcohol (PVA) electrospun nanofibres. Can. J. Microbiol. 2013; 716.
-
Dowlath S, Campbell K, Al-Barwani F, Young VL, Young SL, Walker GF, Ward VK. Dry Formulation of Virus-Like Particles in Electrospun Nanofibers. Vaccines. 2021; 9(3):213.
Open Access
-
Gunderson A, Ramezani M, Orado T K, Monroe M B B. Programming-via-spinning: Electrospun shape memory polymer fibers with simultaneous fabrication and programming. Smart Materials in Medicine 2024; 5: 477.
https://www.sciencedirect.com/science/article/pii/S2590183424000498 Open Access.
-
Guzman J J L, Kara M O P, Frey M W, Angenent L T. Performance of electro-spun carbon nanofiber electrodes with conductive poly(3,4-ethylenedioxythiophene) coatings in bioelectrochemical systems. Journal of Power Sources 2017; 356: 331.
Open Access
-
Lencova S, Stindlova M, Havlickova K, Jencova V, Peroutka V, Navratilova K, Zdenkova K, Stiborova H, Hauzerova S, Kostakova E K, Jankovsky O, Kejzlar P, Lukas D, Demnerova K. Influence of Fiber Diameter of Polycaprolactone Nanofibrous Materials on Biofilm Formation and Retention of Bacterial Cells. ACS Appl. Mater. Interfaces 2024; 16: 25813.
https://pubs.acs.org/doi/full/10.1021/acsami.4c03642 Open Access
-
Lev J, Holba M, Kalhotka L, Mikula P, Kimmer D. Improvements in the Structure of Electrospun Polyurethane Nanofibrous Materials Used for Bacterial Removal from Wastewater. International Journal of Theoretical and Applied Nanotechnology 2012; 1: 16.
Open Access
-
Liyun Zhuo L, Zhang X, Jiang J, Chen H, Liu Y, Wang X, Li W, Zheng G. Electrospinning preparation and characterization testing analysis of nanofiber biofilms. AIP Advances 2024; 14: 025336.
https://pubs.aip.org/aip/adv/article/14/2/025336/3265747 Open Access
-
Liu Y, Rafailovich M H, Malal R, Cohn D, Chidambaram D. Engineering of bio-hybrid materials by electrospinning polymer-microbe fibers. PNAS 2009; 106: 14201.
Open Access
-
Lopez-Rubio A, Sanchez E, Sanz Y, Lagaron J M. Encapsulation of Living Bifidobacteria in Ultrathin PVOH Electrospun Fibers. Biomacromolecules 2009; 10: 2823.
-
Morita Y, Sakamoto H, Suye S I. Characterization of protein adsorption on stretched polyurethane nanofibers prepared by electrospinning. RSC Adv.; 2017: 7 56484.
Open Access
-
Nagy Z K, Wagner I, Suhajda A, Tobak T, Harasztos A H, Vigh T, Soti P L, Pataki H, Molnar K, Marosi G. Nanofibrous solid dosage form of living bacteria prepared by electrospinning. eXPRESS Polymer Letters 2014; 18: 352.
Open Access
-
Salalha W, Kuhn J, Dror Y, Zussman E. Encapsulation of bacteria and viruses in electrospun nanofibres. Nanotechnology 2006; 17: 4675.
-
San N O, Celebioglu A, Tumtas Y, Uqar T, Tekinay T. Reusable bacteria immobilized electrospun nanofibrous webs for decolorization of methylene blue dye in wastewater treatment. RSC Adv. 2014; 4: 32249.
-
Stojanov S, Plavec T V, Zupancic S, Berlec A. Modified vaginal lactobacilli expressing fluorescent and luminescent proteins for more effective monitoring of their release from nanofibers, safety and cell adhesion. Microb Cell Fact 2024; 23: 333.
https://microbialcellfactories.biomedcentral.com/articles/10.1186/s12934-024-02612-w Open Access
-
Stoyanova N, Nachev N, Naydenov M, Valcheva I, Spasova M, Stoilova O. Biocontrol Potential of Poly(3-hydroxybutyrate) Fibers Functionalized with Chitooligosaccharide/Bacillus subtilis Using Electrospinning and Electrospraying. Polymers. 2025; 17(5):692.
https://www.mdpi.com/2073-4360/17/5/692 Open Access.
-
Suaste-Gómez E, Rodríguez-Roldán G, Pérez-Solis I, Torres-Huerta A, Cruz-Cruz C, Tapia-Ramírez J. Electrospinning Polylactic Acid Polymer Membranes as Biological Sieve for Yeast and Bacteria. Materials Sciences and Applications 2022; 13: 389.
Open Access
-
Sugimoto R, Lee J H, Lee J H, Jin H E, Yoo S Y, Lee S W. Bacteriophage nanofiber fabrication using near field electrospinning. RSC Adv. 2019; 9: 39111.
Open Access
-
Tong H W, Mutlu B R, Wackett L P, Aksan A. Silica/PVA biocatalytic nanofibers. Materials Letters 2013; 111: 234.
-
Villarreal-Gomez L J, Vera-Graziano R, Vega-Rios M R, Camacho J L P, Almanza-Reyes H, Mier-Maldonado P A, Cornejo-Bravo J M. Biocompatibility Evaluation of Electrospun Scaffolds of Poly (L-Lactide) with Pure and Grafted Hydroxyapatite. J. Mex. Chem. Soc. 2014; 58: 435.
Open Access
-
Wu X, Li X, Shi Z, Wang X, Wang Z, Li CM. Electrospinning Mo-Doped Carbon Nanofibers as an Anode to Simultaneously Boost Bioelectrocatalysis and Extracellular Electron Transfer in Microbial Fuel Cells. Materials. 2023; 16(6):2479.
Open Access
-
Zhang Q, Lin S, Wang Z, Li J, Xiao J. Lipase-interfacial catalytic systems based on hybrid membranes constructed via electrospinning and gelation. LWT 2023; 183: 114956.
Open Access
-
Zhuo L, Zhang X, Jiang J, Chen H, Zheng Y, Wang X, Li W, Zheng G. Electrospun PMMA fiber biofilm for the removal of COD and NH3-N in wastewater. AIP Advances 2024a; 14: 125005.
https://pubs.aip.org/aip/adv/article/14/12/125005/3323011 Open Access
-
Zupancic S, Skriec K, Kocbek P, Kristl J, Berlec A. Effects of Electrospinning on the Viability of Ten Species of Lactic Acid Bacteria in Poly(Ethylene Oxide) Nanofibers. Pharmaceutics 2019; 11(9): 483.
Open Access
▲ Close list
 ElectrospinTech
ElectrospinTech